  
(E) B-factor representation of the human nCDase crystal structure. (D) Hydrophobic surface representation of human nCDase. (C) Surface representation and surface cutaway of human nCDase highlighting the 20 Å deep, active site pocket. The bound Zn 2+ and Ca 2+ ions are shown as blue and violet spheres, respectively. 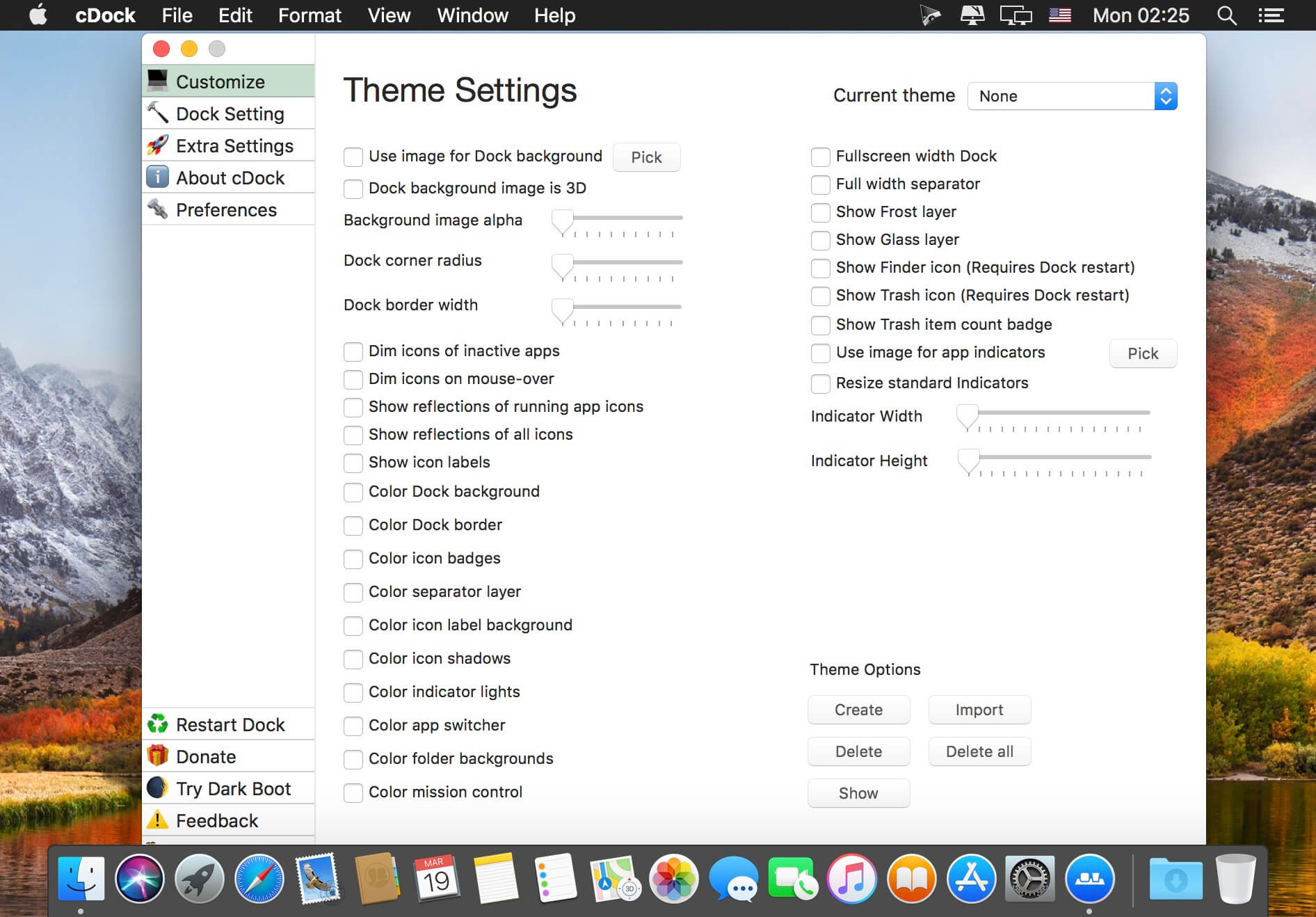
The Asn151-glycan was only observed in one nCDase molecule and is not shown. Asn-linked N-glycans are shown as magenta sticks. Catalytic domain, cyan IG-like domain, yellow domain insertion, orange η2-α8 helices, magenta. (B) Crystal structure of the catalytic and IGlike domains of human nCDase (PDB ID 4WGK). All rights reserved.Ĭrystal structure of human nCDase reveals a novel, 20 Å deep hydrophobic active site pocket (A) Domain organization of human nCDase. Together, these data provide a foundation to aid drug development and establish common themes for how proteins recognize the bioactive lipid ceramide.Ĭopyright © 2015 Elsevier Ltd. Our results suggest that nCDase uses a new catalytic strategy for Zn(2+)-dependent amidases, and generates ceramide specificity by sterically excluding sphingolipids with bulky headgroups and specifically recognizing the small hydroxyl head group of ceramide. Utilizing flexible ligand docking, we predict a likely binding mode for ceramide that superimposes closely with the crystallographically observed transition state analog phosphate. Here, we present the 2.6-Å crystal structure of human nCDase in complex with phosphate that reveals a striking, 20-Å deep, hydrophobic active site pocket stabilized by a eukaryotic-specific subdomain not present in bacterial ceramidases. As an enzyme regulating the balance of ceramide and sphingosine-1-phosphate, nCDase is emerging as a therapeutic target for cancer. Neutral ceramidase (nCDase) catalyzes conversion of the apoptosis-associated lipid ceramide to sphingosine, the precursor for the proliferative factor sphingosine-1-phosphate. 
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
